Imagine waking up one morning and realizing your body is slowly disconnecting from your mind. This is the terrifying reality for people diagnosed with Amyotrophic Lateral Sclerosis, a progressive neurodegenerative disorder commonly known as Lou Gehrig's disease. There is currently no cure, but the landscape of management has changed significantly since the 1990s thanks to a single molecule that altered the course of the disease for thousands of patients. That molecule is Riluzole. Approved by the U.S. Food and Drug Administration (FDA) in May 1995, riluzole was the first pharmacological agent ever authorized specifically for ALS therapy. For decades, it stood alone as the only drug capable of modifying the disease's progression, extending survival time and delaying the critical need for respiratory support.
The Nature of Amyotrophic Lateral Sclerosis
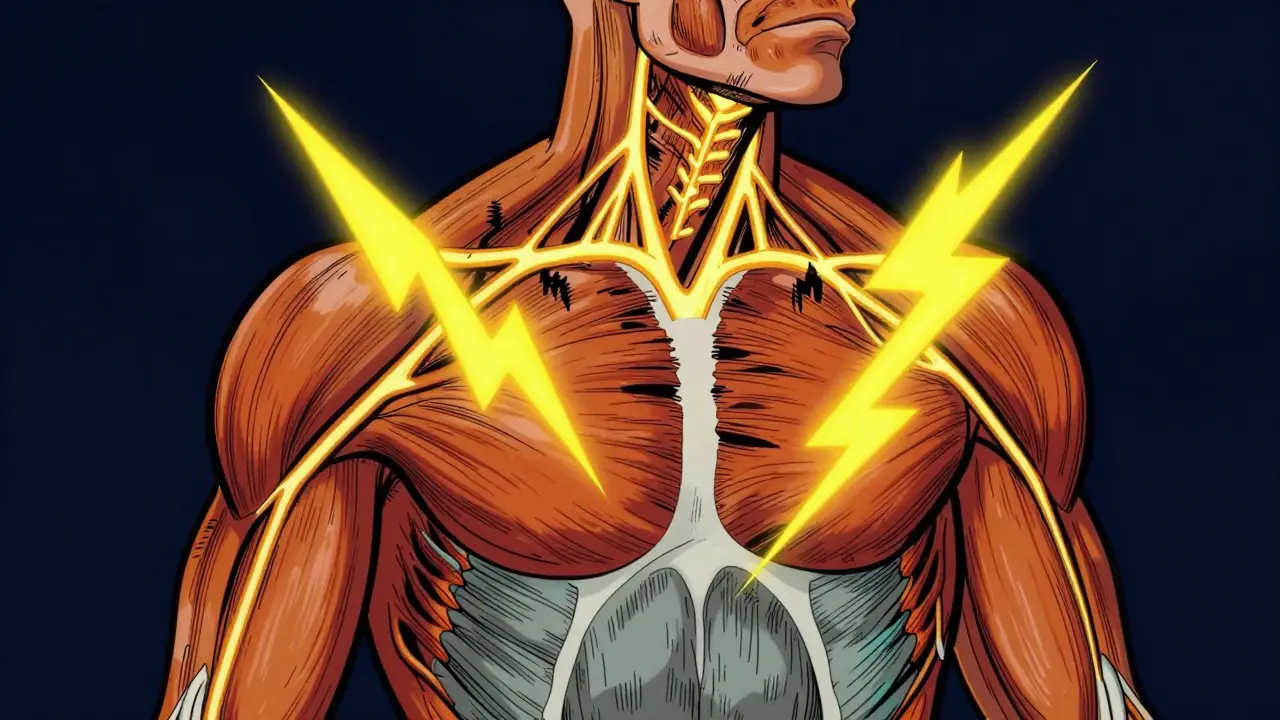
To understand why riluzole matters, you must first grasp what happens inside the nervous system of someone living with ALS. The disease is defined by the selective death of upper and lower motor neurons located in the brain and spinal cord. These neurons are the messengers that carry signals from your brain to your muscles. When they begin to die, the brain's commands are never received. Muscles stop receiving signals, leading to weakness, paralysis, and eventually respiratory failure. Without intervention, the typical progression leads to death within three to five years of symptom onset.
This process isn't just about dying cells; it involves a chemical imbalance. Research suggests that the neurotransmitter Glutamate plays a central role in this destruction. In healthy brains, glutamate helps send electrical signals. In ALS, too much glutamate builds up around the motor neurons. Think of it like a fire hose turned on full blast; the excess fluid damages the pipes. This is known as glutamate excitotoxicity. Overstimulation of NMDA and AMPA receptors triggers cellular damage, essentially causing the neurons to burn out from overwork. This biological mechanism is the precise target of riluzole therapy.
How Riluzole Works to Protect Neurons
Riluzole (C8H8F3N3OS) is a small-molecule benzothiazole compound developed originally by Rhône-Poulenc Rorer, now part of Sanofi. Its exact mode of action has fascinated scientists for over twenty years because it works through multiple pathways simultaneously. According to data from authoritative sources like DrugBank and PubMed, riluzole functions primarily as a Glutamate Antagonist. It blocks the release of glutamate from nerve terminals, effectively turning down the volume of the "fire hose." It achieves this by inactivating voltage-dependent sodium channels on glutamatergic nerves, which prevents the flood of the neurotransmitter into the synapse.
Beyond simply blocking release, riluzole enhances the reuptake of glutamate, clearing the excess from the space between neurons. Additionally, studies indicate it interferes with intracellular signaling events following neurotransmitter binding. A landmark study published in PubMed (ID 8959995) documented that riluzole also blocks the postsynaptic effects of glutamate through noncompetitive antagonism of NMDA receptors. While other drugs targeting similar pathways failed, riluzole succeeded, suggesting its benefits may extend beyond simple glutamate modulation to broader aspects of molecular stability linked to ALS pathogenesis.
Clinical Evidence and Survival Rates
Does the science translate to real life? The pivotal randomized controlled trials provide the evidence we rely on today. Two major studies form the backbone of riluzole's approval. The first trial, published in the New England Journal of Medicine in 1994, showed that a daily dose of 100mg extended median survival by approximately 2 to 3 months compared to placebo. It sounds modest, but in a fatal disease with no cure, every month counts.
A larger, more comprehensive study published in Lancet in 1996 involved 959 patients randomized to receive placebo or varying doses of riluzole for 18 months. The results were statistically significant. Patients taking 100mg daily saw a 35% reduction in the risk of death or tracheostomy compared to those on a placebo. Even higher doses (200mg) showed a 39% reduction, but the adverse event rates were too high, solidifying 100mg as the standard therapeutic dose. Real-world evidence varies slightly. A 2020 analysis of 15 peer-reviewed studies found that roughly half reported a survival extension of 6 to 19 months, while others noted less benefit, highlighting the variability of individual disease progression.
| Metric | Riluzole (100mg/day) | Placebo |
|---|---|---|
| Median Survival Benefit | +2 to 3 Months | Baseline |
| Risk Reduction (Death/Tracheostomy) | 35% | N/A |
| Standard Dosage | 100mg Daily | N/A |
| Patient Continuation Rate | Approx. 63% at 12 Months | N/A |

Forms, Availability, and Usage
Patients have options when it comes to how they take this medication. Originally, riluzole was marketed as Rilutek in 50mg tablets. Taking two pills twice a day is manageable for many, but for patients who have trouble swallowing-a common symptom in advanced ALS-it becomes a hurdle. To address this, the FDA approved new formulations. On October 30, 2018, Tiglutik (oral suspension) became available, allowing the medication to be swallowed easily like a liquid. More recently, on October 6, 2020, Exservan (oral thin film) was approved. This formulation dissolves quickly on the tongue, offering a significant advantage for those with bulbar dysfunction (difficulty speaking or swallowing).
The bioavailability of these formulations differs slightly. The standard tablet has an average oral bioavailability of 60%. However, newer forms like Exservan demonstrated 25% higher bioavailability in clinical trials. This means your body absorbs more of the medicine, potentially stabilizing levels better throughout the day. Regardless of the form, the total daily dose remains consistent at 100mg, split into two 50mg doses. The half-life ranges from 7 to 15 hours, necessitating this twice-daily schedule to maintain protective levels in the bloodstream.
Safety Profile and Managing Side Effects
No powerful drug is without risks, and riluzole requires vigilance. Approximately 10 to 15% of patients discontinue treatment due to adverse effects, according to European Federation of Neurological Societies guidelines. The most common side effect is fatigue, experienced by about 20% of users. Gastrointestinal disturbances are also frequent, with nausea reported in 25% of cases and diarrhea in 15%. Interestingly, taking the medication with food can mitigate some of this nausea.
The most serious concern relates to liver function. Riluzole can elevate liver enzymes. Because of this risk, strict monitoring is mandatory. Standard protocols require liver function tests before starting treatment and monthly checks for the first three months. If enzymes rise above certain thresholds, the doctor may pause or stop the drug. Patients with pre-existing hepatic impairment (Child-Pugh Class B or C) are generally advised to avoid riluzole entirely, as their bodies process the drug differently, leading to dangerously high exposure levels. Conversely, renal impairment does not typically require dose adjustments.

Riluzole in the Broader Treatment Landscape
While riluzole dominated the market alone for over two decades, it is no longer the only option. In May 2017, Edaravone (brand name Radicava) was approved as the second disease-modifying therapy. Edaravone works differently-it acts as a free radical scavenger rather than targeting glutamate directly. Comparisons often arise between the two. Edaravone showed a 33% reduction in functional decline in specific trials, while riluzole focuses on survival time. Many experts now recommend combination therapy, utilizing both agents to target different pathological pathways. In 2023, another breakthrough occurred with the approval of Tofersen for SOD1-mutated ALS, marking the first gene-targeted therapy for a specific subtype. Yet, despite these advances, riluzole remains the foundational pillar of care globally.
The global ALS therapeutics market was valued at $1.27 billion in 2022, with riluzole accounting for roughly 35% of the share. It is estimated that 80-85% of newly diagnosed patients in North America and Europe initiate riluzole treatment. Access remains an issue in low-income regions, where financial barriers limit usage. However, generic versions have helped reduce costs, making the standard of care more accessible than in previous decades.
Future Directions and Patient Perspectives
Patients often weigh the cost of side effects against the potential benefit of buying time. On forums like r/ALS, users share raw experiences. One user noted that after 18 months, their neurologist observed slower progression, attributing this hope to the drug despite managing nausea. Others report frustration when liver issues force discontinuation. Dr. Leonard Petrucelli of the Mayo Clinic predicts that riluzole will remain a cornerstone for the foreseeable future. Emerging research is investigating combining riluzole with other agents like sodium phenylbutyrate to enhance neuroprotection further.
What is the main purpose of taking riluzole?
The primary purpose of riluzole is to slow the progression of muscle weakness caused by Amyotrophic Lateral Sclerosis (ALS). It aims to extend survival by 2-3 months and delay the need for respiratory support, though it does not reverse existing damage or cure the disease.
How long does it take to see benefits from riluzole?
Benefits are not immediate in terms of strength recovery. Peak plasma concentrations are reached within 1-1.5 hours, but observing clinical stabilization takes weeks to months. Most doctors advise continuing treatment regardless of immediate noticeable changes in strength to maximize long-term survival chances.
Can riluzole be stopped suddenly?
You should never stop taking riluzole abruptly without consulting your healthcare provider. Discontinuation can happen due to side effects or liver issues, but doing so without medical supervision might lead to unexpected fluctuations in disease management. Your doctor will guide you on the safety of tapering off.
What foods interact with riluzole?
Caffeine consumption reduces the clearance of riluzole by about 15-20%, meaning caffeine stays in your system longer and might increase side effects. High-fat meals may alter absorption. It is generally recommended to take the medication consistently with or without food as prescribed, but avoiding excessive caffeine intake is wise.
Is riluzole safe for patients with kidney problems?
Yes, generally speaking. Patients with renal impairment do not typically require dose adjustments for riluzole. However, caution is strictly necessary for those with liver impairment, as the drug is metabolized in the liver and toxicity risks increase significantly in those patients.
Comments
Russel Sarong April 1, 2026 at 02:50
Imagine waking up one morning and realizing your body is slowly disconnecting from your mind!!!!!!!! This is the terrifying reality for people diagnosed with Amyotrophic Lateral Sclerosis!!!!!!!!!! There is currently no cure!!!!! But the landscape of management has changed significantly since the 1990s thanks to a single molecule!!!!!!!!!!!! That molecule is Riluzole!!!! Approved by the U.S. Food and Drug Administration in May 1995!!!! riluzole was the first pharmacological agent ever authorized specifically for ALS therapy!!!! For decades it stood alone as the only drug capable of modifying the disease's progression!!!!!!!! It extends survival time and delays the critical need for respiratory support!!!! We cannot ignore the power of science in this fight!!!!!
Julian Soro April 1, 2026 at 16:04
It is really wonderful to see how far we have come with treatment options for motor neuron diseases! Understanding the biological mechanism behind Riluzole gives so much clarity to patients who feel lost in the fog of diagnosis. The fact that it targets glutamate excitotoxicity is a huge step forward in our collective knowledge base. Many people do not realize that taking medication with food can actually help manage the nausea that often accompanies these drugs. It is important to stay consistent with the twice-daily schedule to keep blood levels stable throughout the entire day. Keeping track of liver function tests is also crucial for maintaining safety while on this regimen. Every month of extended life counts for so many families struggling with this condition.
Eleanor Black April 2, 2026 at 06:22
I completely agree with the sentiment regarding the impact of this medication on patient quality of life :) While the survival benefit of two to three months may seem small to the untrained observer it represents significant progress in a field with historically limited options :) It is essential to acknowledge that variability exists in individual disease progression rates despite standardized dosing protocols. The introduction of oral suspensions and thin films has truly revolutionized accessibility for those suffering from bulbar dysfunction. :)
Jenny Gardner April 2, 2026 at 21:59
The new formulations approved in 2018 and 2020 are game changers for compliance!!!!
Owen Barnes April 3, 2026 at 17:23
i think alot of people forget about the side effects part honestly. liver issues can happen quick so monitoring is super important becuz you dont wanna stop the meds unexpectedly. my uncle had to quit after a few months due to enzymes spiking but he said it still helped him longer than expected. its sad when genetics play a bigger role then the druggs can handle sometimes tho.
Callie Bartley April 4, 2026 at 00:38
Oh wow I suppose some people get lucky with the drug tolerance but others get kicked right off the horse. It seems like everyone expects miracles from a medication that only buys a handful of months in the grand scheme of things. Maybe if the pharmaceutical companies focused less on profits and more on actual cures we would not be stuck relying on symptom management tools.
Molly O'Donnell April 4, 2026 at 07:55
The data confirms glutamate excitotoxicity is central to pathology.
Arun Kumar April 6, 2026 at 01:04
We must remember that every patient journey is unique and filled with its own challenges. Supporting the community through shared stories creates a network of hope that transcends medical statistics alone. There is immense value in simply being present for someone navigating this difficult terrain. Small steps forward are victories worth celebrating with genuine enthusiasm and positivity. Let us continue to advocate for broader access to these therapies in low-income regions worldwide. Together we can build a future where no family feels abandoned by the healthcare system.
James DeZego April 7, 2026 at 23:48
Riluzole works primarily as a Glutamate Antagonist in the nervous system :) It blocks the release of glutamate from nerve terminals effectively turning down the volume of the fire hose :) It achieves this by inactivating voltage-dependent sodium channels on glutamatergic nerves :) Beyond simply blocking release riluzole enhances the reuptake of glutamate clearing the excess from the space between neurons :) Additionally studies indicate it interferes with intracellular signaling events following neurotransmitter binding :) A landmark study published in PubMed documented that riluzole also blocks the postsynaptic effects of glutamate through noncompetitive antagonism of NMDA receptors :) While other drugs targeting similar pathways failed riluzole succeeded suggesting its benefits may extend beyond simple glutamate modulation :) This success points toward broader aspects of molecular stability linked to ALS pathogenesis :) The clinical evidence supports the use of daily doses extending median survival by approximately 2 to 3 months :) Real world analyses suggest some patients experience survival extensions ranging from 6 to 19 months :) Patient continuation rates remain high at around 63 percent at twelve months :) New formulations like Tiglutik and Exservan have improved bioavailability for swallowing difficulties :) Bioavailability differences mean your body absorbs more medicine stabilizing levels better throughout the day :) Regardless of the form the total daily dose remains consistent at 100mg split into two 50mg doses :) The half-life ranges from 7 to 15 hours necessitating this twice-daily schedule to maintain protective levels in the bloodstream :) Safety profiles require vigilance as approximately 10 to 15 percent of patients discontinue due to adverse effects :) Liver function monitoring is mandatory monthly for the first three months of treatment :) Patients with hepatic impairment are generally advised to avoid riluzole entirely :) Despite these risks it remains the foundational pillar of care globally today :) Edaravone acts differently as a free radical scavenger rather than targeting glutamate directly :) Many experts now recommend combination therapy utilizing both agents to target different pathological pathways :) Emerging research investigates combining riluzole with other agents like sodium phenylbutyrate to enhance neuroprotection further :)
Rocky Pabillore April 8, 2026 at 06:44
While the layman finds the details satisfactory, one must acknowledge the sophisticated pharmacodynamics at play here. It is imperative that the educated reader distinguishes between mere symptom delay and true disease modification capabilities. The statistical significance of the Lancet study provides a robust foundation for therapeutic justification. We should not diminish the historical context of this compound in favor of newer marketing trends.